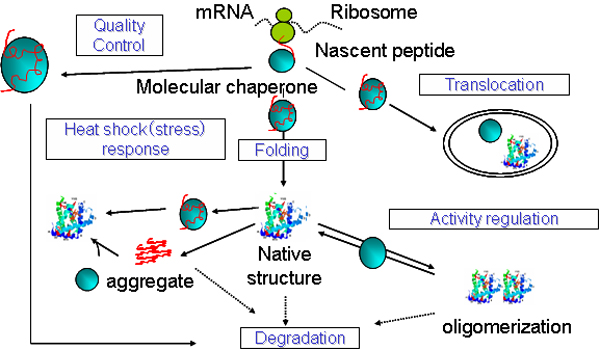
“This coordination probably involves other chaperones besides just UBR7,” Foltz said.Īnn Hogan, PhD, recent graduate from the Driskill Graduate Program in Life Sciences ( DGP), was lead author of the study. In the future, Foltz and his collaborators said they plan to examine other histone chaperone proteins, searching for other dual-use proteins. “It might be a way for the cell to ensure that not only does it have enough building blocks to create DNA, but that it also has enough building blocks to build chromatin,” Foltz said. UBR7 interacts with proteins that form the basic building block of RNA and DNA, raising the possibility it serves a coordinating role for both processes. What makes UBR7 particularly interesting, according to Foltz, is that it already has a previously discovered function: regulating nucleotide metabolism. It’s a memory of the state of the gene before DNA replication.” “The cell has ways to ensure that you can put those modified histones right back, to ensure that information is retained. “If the cell marks a histone and then gets rid of it during replication, then it’s lost that epigenetic information,” Foltz said. Generating cells without UBR7 prevented redeposition of these specific histones. The marker, one that’s associated with gene promoters, is only ever attached to histones that had been packaged into chromatin - making UBR7 among the first histone chaperones known to regulate recycled histones, rather than fresh histones. 
In the current study, Foltz and his collaborators examined the binding of UBR7, finding it bound to histones with a specific epigenetic marker. Histone chaperone proteins ensure that new and old histones - spools around which DNA is looped - are correctly deposited, or recycled and re-deposited, respectively. This requires nucleotide synthesis and packaging of new DNA into chromatin. “We’ve thought of these processes as co-incident but independent, however, this is the first mechanism where we can say they are somehow coupled,” Foltz said.Ĭells undergoing proliferation - dividing and replicating- must replicate their DNA to provide a genetic template for the new cell. This protein was previously identified to regulate nucleotide metabolism, making UBR7 among the first proteins known to affect both processes, according to Daniel Foltz, PhD, associate professor of Biochemistry and Molecular Genetics, and senior author of the study. The protein UBR7 acts as a histone chaperone, regulating histone re-deposition at specific sites during DNA replication, according a recent study published in The EMBO Journal. Daniel Foltz, PhD, associate professor of Biochemistry and Molecular Genetics, was senior author of the study published in The EMBO Journal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
